How Turmeric Reacts Chemically with Acids and Bases
Turmeric, commonly known as haldi, is a bright yellow spice obtained from the rhizome of the plant Curcuma longa. It has been used for centuries in cooking, traditional medicine, and natural remedies. Apart from its medicinal importance, turmeric also has interesting chemical properties that make it useful in simple scientific experiments. One of the most fascinating aspects of turmeric chemistry is how it reacts with acids and bases. These reactions are mainly due to the presence of its active compound called curcumin, which changes color when it interacts with different chemical environments.
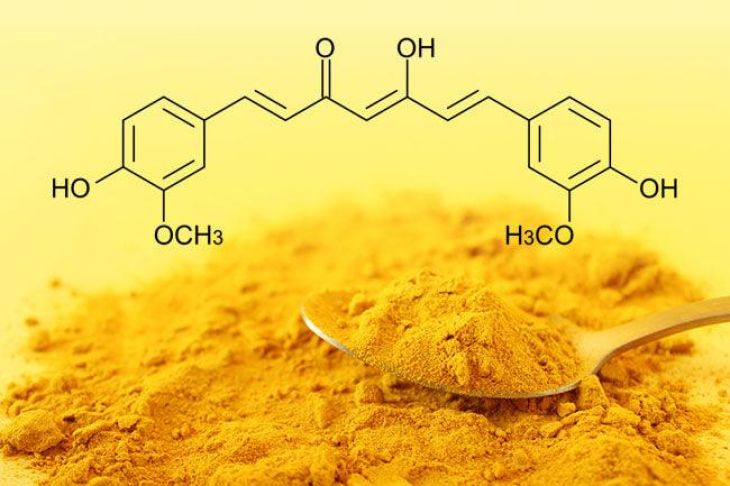
Curcumin is a natural polyphenolic compound responsible for turmeric’s color and many of its biological activities. Chemically, curcumin contains several functional groups, including phenolic hydroxyl groups and conjugated double bonds. These structural features allow the molecule to interact with acids, bases, and other chemical substances. Because of this property, turmeric is sometimes used as a natural pH indicator, meaning it can help identify whether a solution is acidic or basic.
To understand turmeric’s reaction with acids and bases, it is first important to understand the concept of pH. The pH scale measures how acidic or basic a substance is. Substances with a pH below 7 are considered acidic, while those with a pH above 7 are basic or alkaline. Neutral substances have a pH of around 7. Many natural indicators change color depending on the pH of the environment, and turmeric is one such indicator.
In its natural state, turmeric powder has a bright yellow color. When it comes into contact with acidic solutions, such as lemon juice or vinegar, it usually retains its yellow appearance. This happens because curcumin molecules remain relatively stable in acidic conditions. The chemical structure of curcumin does not change significantly when exposed to mild acids, so the color remains mostly unchanged.
However, when turmeric interacts with basic or alkaline substances, a noticeable chemical reaction occurs. In alkaline conditions, the curcumin molecule undergoes a structural change that alters how it absorbs and reflects light. As a result, the yellow color of turmeric can turn reddish-brown or deep orange when it reacts with a base. This color change is one of the reasons turmeric is widely used in school experiments and demonstrations to show acid–base reactions.
For example, if turmeric powder is mixed with baking soda or soap solution, the color often changes from yellow to a reddish or brownish shade. These substances are basic in nature, and they trigger a chemical transformation in the curcumin molecule. The reaction occurs because the phenolic groups in curcumin lose hydrogen ions in alkaline conditions, forming a different chemical structure that absorbs light differently.
The reaction of turmeric with acids and bases is not only visually interesting but also scientifically important. It demonstrates how natural compounds respond to environmental changes at the molecular level. This property has practical applications in chemistry education and in the development of natural indicators.
Key Chemical Behaviors of Turmeric in Acidic and Basic Conditions
The chemical reactions of turmeric with acids and bases can be summarized through several important observations:
-
Stable yellow color in acidic solutions, such as lemon juice or vinegar.
Color change to reddish-brown in alkaline solutions, such as soap or baking soda. -
Curcumin acts as a natural pH indicator, helping distinguish between acidic and basic substances.
Phenolic groups in curcumin react with bases, leading to structural changes in the molecule. -
Acidic environments maintain the original structure of curcumin, preventing major color change.
-
Alkaline environments cause deprotonation, which alters the electronic structure of curcumin.
One practical example of this reaction is the turmeric paper test, which is a simple experiment used in chemistry laboratories and classrooms. In this test, turmeric powder is mixed with alcohol or water to create a solution. Filter paper is then soaked in this solution and allowed to dry. The resulting turmeric-coated paper can be used to detect alkaline substances. When the paper comes into contact with a base, it changes color from yellow to reddish-brown, indicating the presence of an alkaline compound.
This natural indicator is particularly useful because turmeric is inexpensive, safe, and widely available. Unlike some synthetic indicators that require specialized chemicals, turmeric can be easily prepared at home or in a classroom setting. For this reason, it is often used as a teaching tool to introduce students to acid–base chemistry.
The chemical behavior of turmeric also has relevance in food science. Many foods contain natural acids or bases that may influence the color of turmeric during cooking. For instance, when turmeric is added to dishes containing alkaline ingredients, slight color changes may occur. However, these changes are usually subtle and do not significantly affect the taste or nutritional value of the food.
Turmeric’s chemical response to pH changes also reflects the versatility of natural compounds found in plants. Many plant pigments and bioactive molecules are sensitive to environmental conditions such as temperature, light, and acidity. These properties help scientists understand how natural molecules function and how they can be used in various applications.
Researchers have also explored the possibility of using turmeric-based compounds in advanced chemical sensors. Because curcumin responds to changes in pH and other chemical conditions, it may serve as a component in environmentally friendly detection systems. For example, scientists are studying how natural dyes like curcumin could be used in smart packaging materials that detect food spoilage by changing color.
Another interesting aspect of turmeric chemistry is its stability under different conditions. While curcumin remains relatively stable in acidic environments, it can degrade more quickly in strong alkaline solutions. When exposed to high pH levels, the molecule may break down into smaller compounds such as ferulic acid and vanillin. These degradation reactions highlight the importance of pH in determining the chemical behavior of natural compounds.
Understanding how turmeric reacts with acids and bases also helps scientists study the broader chemical properties of curcumin. By examining how its molecular structure changes under different conditions, researchers can learn more about its reactivity, stability, and potential applications in medicine, chemistry, and food science.
In conclusion, turmeric exhibits fascinating chemical behavior when exposed to acidic and basic environments. The active compound curcumin remains stable and yellow in acidic conditions but undergoes a structural transformation in alkaline solutions, producing a reddish-brown color. This property allows turmeric to function as a natural pH indicator and provides a simple way to demonstrate acid–base reactions. Beyond classroom experiments, the chemical interactions of turmeric highlight the complexity and versatility of natural compounds found in plants. As scientific research continues to explore these properties, turmeric remains not only a valuable spice and medicinal herb but also an intriguing subject in the study of natural chemistry.