Turmeric as a Natural Chemical Indicator in Acid–Base Reactions
Turmeric, commonly known as haldi, is a bright yellow spice widely used in cooking, traditional medicine, and cultural practices. It is obtained from the rhizome of the plant Curcuma longa and has been valued for centuries for its medicinal and nutritional properties. Besides its importance in food and health, turmeric also has interesting chemical characteristics that make it useful in scientific experiments. One such property is its ability to act as a natural chemical indicator in acid–base reactions.
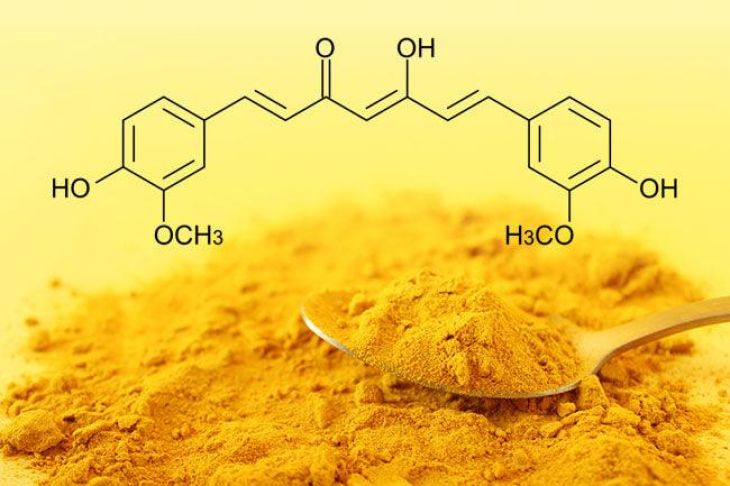
In chemistry, indicators are substances that change color depending on the acidity or basicity of a solution. They help scientists determine whether a substance is acidic, basic, or neutral. Many commonly used indicators, such as litmus or phenolphthalein, are synthetic chemicals. However, nature also provides several substances that behave in a similar way. Turmeric is one of the best-known natural indicators, and its color-changing ability is mainly due to its active compound called curcumin.
Curcumin is the pigment responsible for the bright yellow color of turmeric. Chemically, it belongs to a class of compounds known as polyphenols. The molecular structure of curcumin contains functional groups that are sensitive to changes in pH, which is a measure of acidity or alkalinity. Because of this property, curcumin reacts differently when it is exposed to acidic or basic substances.
The pH scale ranges from 0 to 14. Substances with a pH below 7 are considered acidic, while those with a pH above 7 are basic or alkaline. Neutral substances, such as pure water, have a pH of around 7. Indicators help identify where a substance falls on this scale by producing visible color changes when they react with different pH levels.
In its natural form, turmeric has a strong yellow color. When turmeric is placed in an acidic solution, such as lemon juice or vinegar, it usually retains its yellow color. This happens because the curcumin molecules remain stable in acidic conditions. The structure of the molecule does not undergo significant chemical changes, so the color remains mostly the same.
However, when turmeric is exposed to a basic or alkaline substance, the situation changes. In alkaline environments, curcumin molecules undergo a chemical reaction that alters their structure. This change affects the way the molecules absorb and reflect light, resulting in a visible color shift. As a result, turmeric changes from yellow to reddish-brown or dark orange when it reacts with bases.
This color change makes turmeric a useful natural indicator for detecting basic substances. For example, when turmeric comes into contact with soap solution, baking soda, or certain cleaning agents, its color changes quickly, indicating that the substance is alkaline. Because of this property, turmeric can be used in simple experiments to demonstrate acid–base reactions.
Key Properties That Make Turmeric a Natural Indicator
Turmeric functions effectively as a natural indicator because of several chemical and physical properties:
-
Presence of curcumin, a pH-sensitive compound responsible for color change.
-
Stable yellow color in acidic solutions, such as vinegar or lemon juice.
Color transformation to reddish-brown in alkaline conditions, such as soap or baking soda. -
Natural and non-toxic composition, making it safe for educational experiments.
-
Easily available and inexpensive, allowing simple preparation at home or in classrooms.
-
Clear and visible color change, which helps identify basic substances quickly.
One of the most common applications of turmeric as an indicator is the turmeric paper test. This is a simple method used in chemistry laboratories and classrooms to detect alkaline substances. The test is prepared by making a solution of turmeric powder mixed with water or alcohol. Strips of filter paper are soaked in this solution and then dried. The dried strips become turmeric indicator paper.
When these turmeric-coated paper strips are touched to a solution, they can reveal whether the substance is alkaline. If the strip changes from yellow to reddish-brown, it indicates the presence of a base. If there is no change in color, the solution is either acidic or neutral. This simple test helps students understand how natural indicators work in chemical reactions.
The reason turmeric changes color in alkaline conditions is related to the behavior of the phenolic groups present in the curcumin molecule. In basic environments, these groups lose hydrogen ions, a process known as deprotonation. This chemical change modifies the electronic structure of the molecule, causing it to absorb light differently and produce a new color. In contrast, acidic environments maintain the original molecular structure of curcumin, preventing the color change.
Turmeric’s role as a natural indicator highlights the connection between chemistry and everyday materials. Many plants contain pigments that respond to changes in pH. For example, red cabbage, beetroot, and certain flowers also exhibit color changes when exposed to acids and bases. These natural indicators provide environmentally friendly alternatives to synthetic chemicals.
The use of turmeric as an indicator is especially valuable in educational settings. Because turmeric is safe, inexpensive, and widely available, teachers often use it to demonstrate acid–base reactions in classrooms. Students can easily perform experiments using household materials such as lemon juice, baking soda, and soap solution. These simple demonstrations help learners understand fundamental chemical concepts in an engaging way.
In addition to education, turmeric-based indicators have attracted interest in scientific research. Scientists are exploring the possibility of using natural pigments like curcumin in environmentally friendly chemical sensors. Such sensors could detect changes in pH in food, water, or industrial processes. Because curcumin is biodegradable and non-toxic, it offers a sustainable alternative to synthetic indicators.
Another advantage of turmeric indicators is their visual clarity. The contrast between bright yellow and reddish-brown is easy to observe, even without specialized laboratory equipment. This makes turmeric particularly useful in situations where simple and quick identification of alkaline substances is required.
Despite its usefulness, turmeric indicators also have certain limitations. For instance, turmeric does not show a strong color change in acidic solutions. It mainly reacts to alkaline conditions, meaning it is more suitable for detecting bases than acids. In addition, very strong bases may cause the color to fade or degrade over time due to chemical breakdown of curcumin.
In conclusion, turmeric is not only an important spice and medicinal plant but also a fascinating natural chemical indicator. Its active compound curcumin responds to changes in pH, allowing turmeric to change color in alkaline environments. This property enables turmeric to be used in simple acid–base experiments, particularly in detecting basic substances. Because it is safe, natural, and easily available, turmeric serves as an excellent educational tool for demonstrating chemical reactions. The study of turmeric as an indicator also highlights the broader potential of plant-based compounds in scientific applications and environmentally friendly technologies.