Chemical Properties of Curcumin: Structure, Solubility, and Stability
Curcumin is the principal bioactive compound found in turmeric, the bright yellow spice obtained from the rhizome of the plant Curcuma longa. It is responsible for many of turmeric’s well-known biological and medicinal properties, including its antioxidant, anti-inflammatory, and potential therapeutic effects. Because of these benefits, curcumin has attracted significant attention in the fields of chemistry, pharmacology, and medical research. To understand how curcumin works in the body and why it is sometimes difficult to use as a drug, it is important to examine its chemical properties, particularly its molecular structure, solubility, and stability.
Curcumin belongs to a class of natural compounds known as polyphenols. Polyphenols are plant-derived molecules that contain multiple phenolic groups and are widely recognized for their ability to act as antioxidants. These compounds are commonly found in fruits, vegetables, spices, and herbs. Curcumin stands out among polyphenols because of its distinctive chemical structure and vibrant yellow color, which makes it a natural dye as well as a medicinal compound.
Molecular Structure of Curcumin
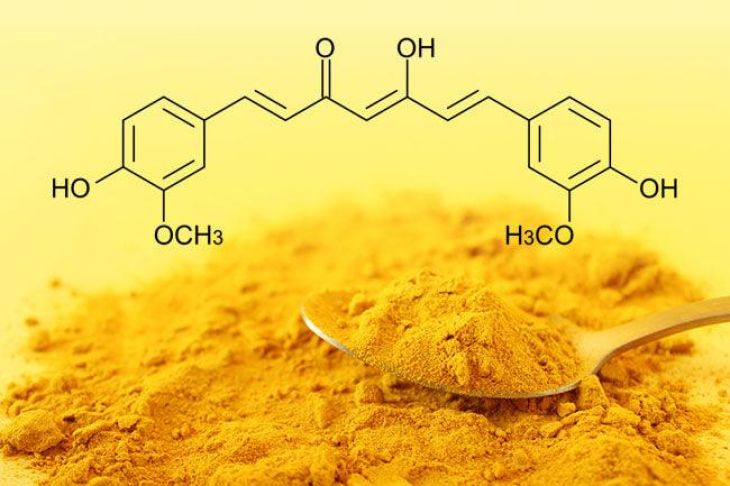
The chemical structure of curcumin is one of the key reasons for its unique biological activity. Curcumin has the molecular formula C₂₁H₂₀O₆ and is classified as a diarylheptanoid. This means the molecule contains two aromatic rings connected by a seven-carbon chain. The aromatic rings are phenolic in nature, meaning they contain hydroxyl (-OH) groups that contribute to curcumin’s antioxidant properties.
Between these aromatic rings lies a central chain that includes conjugated double bonds and a diketone functional group. The presence of these conjugated bonds allows electrons to move freely within the molecule, which is responsible for curcumin’s strong color and chemical reactivity. This electron movement also enables curcumin to interact with various molecules in the body, including free radicals and enzymes.
Another interesting feature of curcumin’s structure is its ability to exist in two chemical forms known as keto and enol forms. This phenomenon is called tautomerism. In acidic and neutral environments, the keto form is generally more stable, while in alkaline conditions, the enol form tends to dominate. The ability to switch between these forms influences how curcumin behaves in different chemical environments.
Solubility of Curcumin
One of the major chemical challenges associated with curcumin is its poor solubility in water. Curcumin is considered a hydrophobic compound, meaning it does not dissolve easily in aqueous solutions. This property significantly affects how curcumin is absorbed and utilized by the human body.
In contrast to its low water solubility, curcumin dissolves more readily in organic solvents such as ethanol, methanol, acetone, and certain oils. Because of this, curcumin is often extracted from turmeric using organic solvents during laboratory or industrial processing. The hydrophobic nature of curcumin also explains why it is often consumed in foods that contain fats or oils, which can improve its absorption in the digestive system.
Low solubility is one of the main reasons curcumin has limited bioavailability. Bioavailability refers to the proportion of a substance that enters the bloodstream and becomes available for biological activity. When curcumin is consumed in its natural form, only a small fraction is absorbed into the body, while the rest passes through the digestive system. Scientists are actively researching methods to improve curcumin’s solubility and absorption, including the development of nanoparticles, liposomal formulations, and combinations with other compounds.
Stability of Curcumin
Curcumin’s chemical stability is another important factor that influences its effectiveness in food, medicine, and research applications. Stability refers to how well a compound maintains its chemical structure under different environmental conditions such as light, heat, and pH.
Curcumin is relatively stable in acidic and neutral environments but becomes unstable under alkaline conditions. When exposed to alkaline solutions, curcumin may undergo chemical degradation, breaking down into smaller molecules such as ferulic acid and vanillin. This instability can limit its effectiveness in certain pharmaceutical formulations or laboratory experiments.
Light exposure also affects curcumin stability. Curcumin is sensitive to ultraviolet and visible light, which can cause photodegradation. When exposed to strong light for extended periods, the molecule may lose its color and break down into other chemical compounds. This is why curcumin-containing products are often stored in dark or opaque containers to reduce light exposure.
Temperature plays a role as well. While curcumin can tolerate moderate heat, prolonged exposure to high temperatures may gradually degrade the compound. However, typical cooking temperatures used in food preparation generally do not completely destroy curcumin, allowing it to retain much of its beneficial activity.
Key Chemical Characteristics of Curcumin
Several chemical features define the behavior and applications of curcumin:
-
Polyphenolic structure with two aromatic rings and hydroxyl groups.
-
Conjugated double bonds that give curcumin its bright yellow color.
-
Keto–enol tautomerism allowing the molecule to exist in two structural forms.
-
Hydrophobic nature, resulting in low water solubility.
-
Higher solubility in organic solvents and oils.
-
Sensitivity to alkaline pH, which can cause chemical degradation.
-
Light sensitivity, leading to photodegradation when exposed to strong light.
Influence of Chemical Properties on Biological Activity
The chemical characteristics of curcumin directly influence its biological behavior. The phenolic hydroxyl groups and conjugated double bonds allow curcumin to neutralize free radicals and reduce oxidative stress. These antioxidant effects are thought to contribute to many of curcumin’s health benefits, including protection against inflammation and cellular damage.
At the same time, the compound’s poor solubility and instability in certain conditions present challenges for its use as a therapeutic drug. Scientists are therefore exploring ways to modify curcumin chemically or deliver it through advanced drug delivery systems that enhance its stability and absorption.
Nanotechnology has become a promising area of research for curcumin delivery. Encapsulating curcumin in nanoparticles or liposomes can improve its solubility, protect it from degradation, and increase its bioavailability. Such innovations may help unlock the full potential of curcumin in medical applications.
Conclusion
Curcumin is a chemically fascinating compound with unique structural features that give rise to its vibrant color and biological activity. Its molecular structure includes aromatic rings, conjugated double bonds, and functional groups that allow it to act as a powerful antioxidant. However, curcumin’s chemical behavior also presents challenges, particularly its low water solubility and sensitivity to environmental conditions such as light and alkaline pH. Understanding these chemical properties is essential for researchers seeking to enhance curcumin’s effectiveness in food, medicine, and pharmaceutical applications. As scientific research continues to advance, improved formulations and technologies may help overcome these limitations, enabling curcumin to play an even greater role in health and therapeutic development.