Chemical Behavior of Turmeric Pigments at Different pH Levels
Turmeric, a widely used spice derived from the rhizome of the Curcuma longa plant, is well known for its vibrant yellow color and numerous health benefits. The pigment responsible for this characteristic color is primarily curcumin, a natural polyphenolic compound. Beyond its culinary and medicinal uses, curcumin has attracted scientific interest due to its fascinating chemical behavior, particularly its response to changes in pH. Understanding how turmeric pigments behave in acidic, neutral, and alkaline environments provides valuable insights into their applications in food science, chemistry, and even industrial processes.
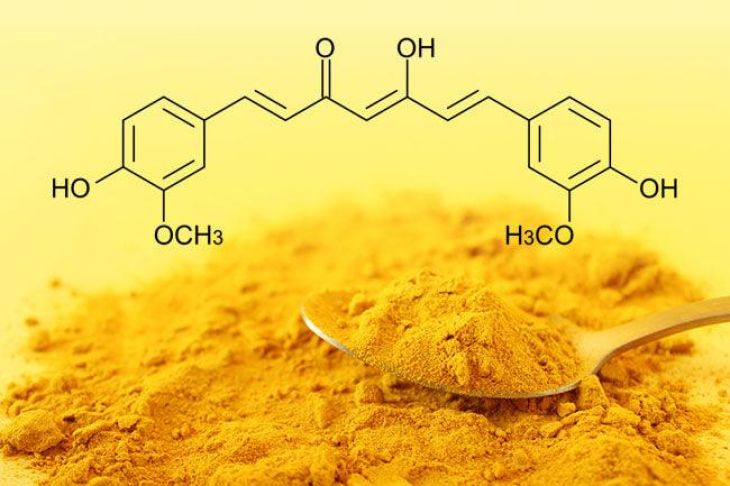
Curcumin is highly sensitive to pH variations, and its color changes significantly depending on the acidity or alkalinity of its environment. This property makes it not only an important natural coloring agent but also a useful pH indicator in certain contexts. The molecular structure of curcumin contains phenolic groups and a conjugated system of double bonds, both of which influence how the molecule interacts with hydrogen ions (H⁺) in different conditions.
In acidic conditions, where the pH is low, curcumin generally maintains its bright yellow color. This is because the molecular structure remains relatively stable, with minimal ionization of its functional groups. The phenolic hydroxyl groups in curcumin are less likely to lose hydrogen ions in such environments, preserving the original structure and color of the pigment. This is why turmeric retains its golden-yellow appearance in dishes that contain acidic ingredients such as lemon juice or tamarind.
As the pH moves toward neutral, curcumin continues to exhibit a yellow to slightly orange hue. At this stage, the molecule remains relatively stable, although slight changes in its structure may begin to occur. The conjugated system of double bonds still allows for efficient light absorption, which is responsible for its visible color. Most everyday cooking conditions fall within this pH range, making turmeric a reliable coloring agent in a variety of dishes.
However, the most noticeable changes in curcumin’s behavior occur in alkaline conditions, where the pH is high. In such environments, curcumin undergoes deprotonation, meaning that it loses hydrogen ions from its phenolic groups. This leads to a change in the electronic structure of the molecule, altering how it absorbs light. As a result, the color of turmeric shifts from bright yellow to a reddish-brown or deep orange shade. This dramatic transformation is a clear indication of curcumin’s sensitivity to pH changes.
Key observations of turmeric pigment behavior at different pH levels include:
-
Acidic conditions (low pH): Bright yellow color remains stable
-
Neutral conditions (pH ~7): Yellow to light orange color with minimal change
-
Alkaline conditions (high pH): Color shifts to reddish-brown or deep orange
-
Ionization effects: Loss or gain of hydrogen ions alters molecular structure
-
Reversible changes: Some color changes may reverse when pH is adjusted
-
Indicator potential: Can act as a natural pH indicator in experiments
The ability of curcumin to change color with pH has practical applications beyond the kitchen. In chemistry laboratories, turmeric extract can be used as a natural pH indicator. For example, when turmeric paper is exposed to a basic substance such as soap or baking soda solution, it changes from yellow to reddish-brown. This simple yet effective demonstration is often used in educational settings to illustrate acid-base reactions.
Another important aspect of curcumin’s chemical behavior is its stability. While it remains relatively stable in acidic and neutral conditions, it tends to degrade more rapidly in alkaline environments. The breakdown of curcumin can lead to the formation of smaller compounds, which may not retain the same color or biological activity. This instability is an important consideration in food processing and pharmaceutical applications, where maintaining the integrity of the compound is crucial.
Temperature and light also interact with pH to influence the behavior of turmeric pigments. High temperatures and prolonged exposure to light can accelerate the degradation of curcumin, especially in alkaline conditions. This is why turmeric-containing foods are often cooked carefully and stored in conditions that minimize exposure to heat and light.
The pH-dependent behavior of curcumin also plays a role in its biological activity. In the human body, different tissues and environments have varying pH levels. For instance, the stomach is highly acidic, while the intestines are more neutral to slightly alkaline. These variations can affect how curcumin is absorbed, metabolized, and utilized in the body. Understanding these interactions is important for optimizing the use of turmeric in medical and nutritional applications.
In the food industry, the sensitivity of turmeric pigments to pH is both an advantage and a challenge. On one hand, it allows for the creation of visually dynamic foods and beverages. On the other hand, it requires careful formulation to ensure consistent color and stability. Food scientists must consider factors such as ingredient composition, processing conditions, and storage environment to maintain the desired appearance and quality of turmeric-based products.
Additionally, the natural origin of turmeric pigments makes them an attractive alternative to synthetic dyes, especially in the context of clean-label products. However, their sensitivity to pH and environmental factors means that they must be used thoughtfully to achieve optimal results.
In conclusion, the chemical behavior of turmeric pigments at different pH levels is a fascinating example of how molecular structure influences physical properties. From maintaining a bright yellow color in acidic conditions to shifting to reddish-brown in alkaline environments, curcumin demonstrates remarkable versatility. These changes are driven by ionization and alterations in the molecule’s electronic structure, highlighting the intricate relationship between chemistry and color. Understanding these properties not only enhances the use of turmeric in cooking but also opens up possibilities in scientific, industrial, and medical applications.