Chemical Comparison Between Curcumin and Vitamin B12
Curcumin and vitamin B12 are two biologically important compounds that play very different roles in human health, yet both are widely studied in chemistry and medicine. Curcumin, derived from turmeric (Curcuma longa), is a natural polyphenol known for its anti-inflammatory and antioxidant properties. Vitamin B12, also known as cobalamin, is an essential water-soluble vitamin required for nerve function, red blood cell formation, and DNA synthesis. Although both compounds contribute to health, their chemical structures, properties, and biological roles differ significantly. A comparative analysis of curcumin and vitamin B12 highlights the diversity of naturally occurring molecules and their unique mechanisms of action.
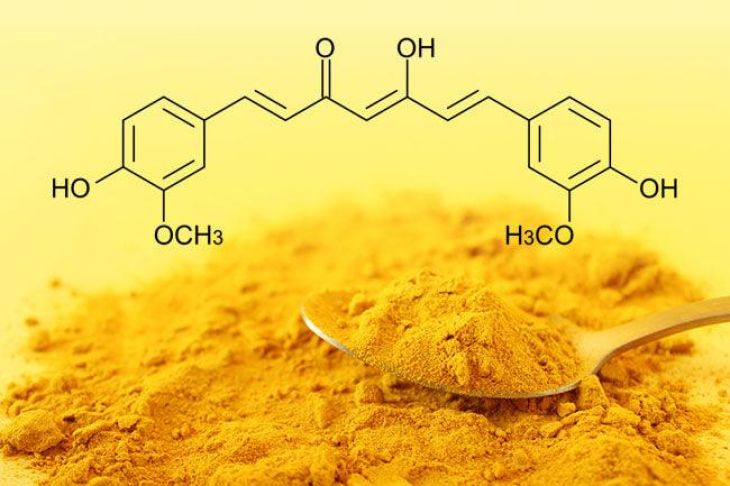
From a structural perspective, curcumin and vitamin B12 are fundamentally different. Curcumin is a relatively small organic molecule with the chemical formula C₂₁H₂₀O₆. It consists of two aromatic rings connected by a conjugated chain containing a β-diketone group. This structure is symmetrical and rich in functional groups such as phenolic hydroxyl (-OH) and methoxy (-OCH₃) groups. These features contribute to its antioxidant behavior and its ability to interact with free radicals and metal ions.
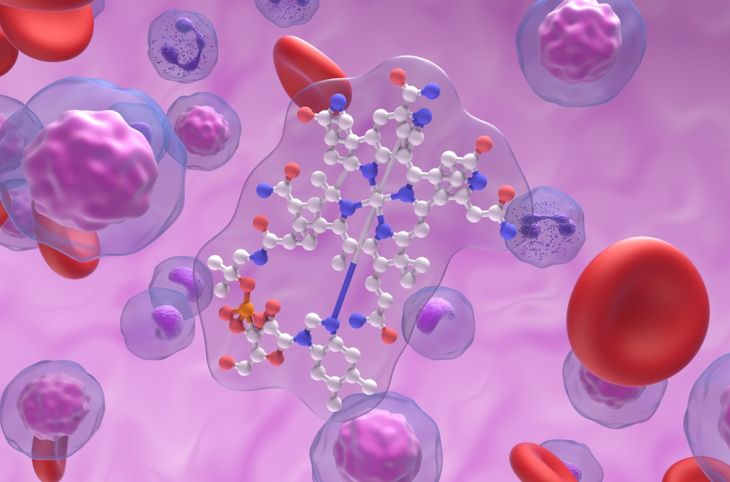
In contrast, vitamin B12 is one of the most structurally complex vitamins known. Its chemical structure is based on a large corrin ring system that surrounds a central cobalt ion. The presence of this metal ion makes vitamin B12 unique among vitamins. The cobalt atom can form different bonds with various ligands, resulting in different forms of vitamin B12, such as methylcobalamin and cyanocobalamin. This complexity allows vitamin B12 to participate in critical biochemical reactions in the body.
Another major difference lies in their solubility and chemical behavior. Curcumin is hydrophobic, meaning it does not dissolve well in water but is soluble in fats and organic solvents. This property affects its absorption in the body, leading to relatively low bioavailability. On the other hand, vitamin B12 is water-soluble and can dissolve easily in aqueous environments. However, despite its solubility, its absorption in the body is a complex process that requires intrinsic factor, a protein produced in the stomach.
The functional groups present in each compound also influence their chemical properties and biological activities. Curcumin’s phenolic and diketone groups enable it to act as an antioxidant and a metal chelator. These groups can donate electrons or hydrogen atoms, neutralizing free radicals and reducing oxidative stress. Vitamin B12, in contrast, contains a cobalt-centered coordination complex that allows it to participate in enzymatic reactions. It acts as a cofactor in processes such as DNA synthesis and the metabolism of fatty acids and amino acids.
Key chemical differences between curcumin and vitamin B12 include:
-
Molecular size: Curcumin is a small organic molecule, while vitamin B12 is large and complex
-
Core structure: Curcumin has a linear polyphenolic structure; vitamin B12 has a corrin ring with a cobalt ion
-
Solubility: Curcumin is fat-soluble; vitamin B12 is water-soluble
-
Functional groups: Curcumin contains phenolic and diketone groups; vitamin B12 contains a metal-centered coordination complex
-
Biological role: Curcumin acts mainly as an antioxidant; vitamin B12 functions as a coenzyme
-
Source: Curcumin is plant-derived; vitamin B12 is obtained from animal-based foods or microbial synthesis
-
Stability: Curcumin is sensitive to light and pH; vitamin B12 is sensitive to heat and light but generally more stable in physiological conditions
The chemical stability of these compounds further distinguishes them. Curcumin is relatively unstable under certain conditions, particularly in alkaline environments or when exposed to light and oxygen. It can degrade into smaller compounds, reducing its effectiveness. Vitamin B12, while also sensitive to light and heat, is generally more stable within the body due to its complex structure and binding with proteins.
In terms of biological function, the differences are even more pronounced. Curcumin is not an essential nutrient, meaning the body does not require it for survival. However, it provides beneficial effects by modulating inflammation, oxidative stress, and cellular signaling pathways. Its role is more supportive and protective rather than fundamental.
Vitamin B12, on the other hand, is essential for human health. It plays a critical role in the formation of red blood cells, maintenance of the nervous system, and synthesis of DNA. A deficiency in vitamin B12 can lead to serious health issues, including anemia and neurological disorders. This highlights the importance of its unique chemical structure, which enables it to function as a coenzyme in vital metabolic processes.
Another interesting aspect of comparison is their interaction with metals. Curcumin can bind to metal ions through its functional groups, acting as a chelating agent. This property contributes to its antioxidant activity. Vitamin B12, however, already contains a metal ion (cobalt) at its core, which is essential for its biological function. Rather than binding external metals, it uses its central cobalt atom to facilitate biochemical reactions.
The methods of absorption and metabolism of these compounds also differ significantly. Curcumin’s low water solubility limits its absorption in the digestive tract, although combining it with substances like black pepper can enhance its bioavailability. Vitamin B12 requires a specialized absorption mechanism involving intrinsic factor and receptors in the small intestine, making its uptake more complex but highly regulated.
From a pharmaceutical perspective, both compounds are of great interest but for different reasons. Curcumin is studied for its potential therapeutic effects in conditions such as inflammation, cancer, and neurodegenerative diseases. Researchers are working to improve its stability and absorption to make it more effective as a բուժing agent. Vitamin B12, meanwhile, is widely used in clinical medicine to treat deficiencies and related disorders, often administered as injections or supplements.
In conclusion, curcumin and vitamin B12 represent two distinct classes of biologically important compounds with vastly different chemical structures and functions. Curcumin, a polyphenolic compound from turmeric, is valued for its antioxidant and anti-inflammatory properties, while vitamin B12, a cobalt-containing vitamin, is essential for critical metabolic processes. Their differences in molecular structure, solubility, stability, and biological roles highlight the diversity of natural compounds and their unique contributions to human health. Understanding these differences not only deepens our knowledge of chemistry but also underscores the importance of both compounds in their respective domains.