Interaction of Curcumin with Metal Ions
Curcumin, the principal bioactive compound derived from turmeric, has gained considerable attention in scientific research due to its wide range of biological and chemical properties. Known for its antioxidant, anti-inflammatory, and therapeutic potential, curcumin also exhibits an important chemical characteristic—its ability to interact with metal ions. This interaction, commonly referred to as metal chelation, plays a significant role in both its biological activity and its potential applications in medicine, environmental science, and material chemistry.
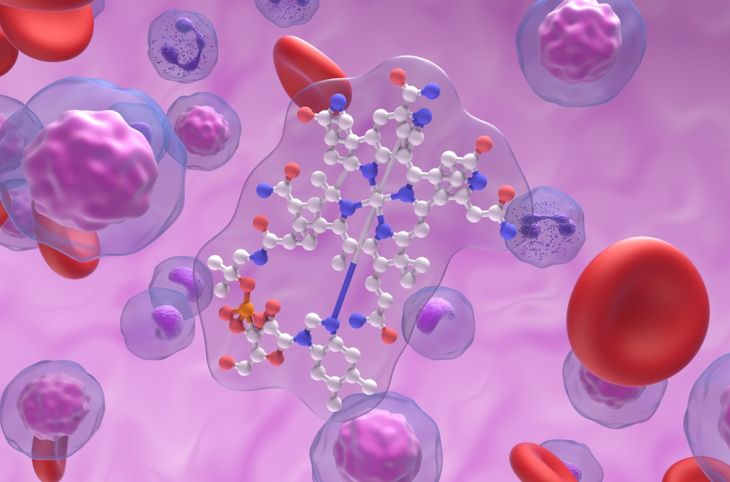
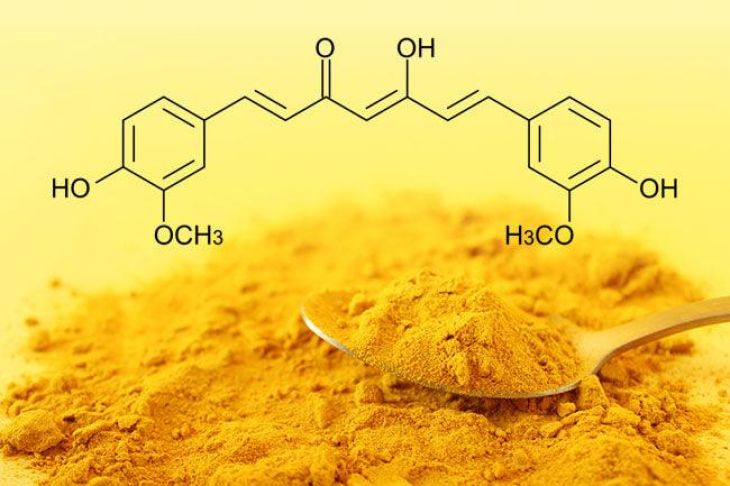
At the molecular level, curcumin possesses a unique structure that allows it to bind with various metal ions. Its chemical framework includes a β-diketone moiety and phenolic hydroxyl groups, both of which act as active sites for binding. These functional groups can donate electrons to metal ions, forming stable complexes. This property enables curcumin to interact with metals such as iron, copper, zinc, and aluminum, among others.
The ability of curcumin to chelate metal ions is particularly important in biological systems. Metal ions are essential for many physiological processes, including enzyme activity, oxygen transport, and cellular signaling. However, excess metal ions, especially transition metals like iron and copper, can contribute to the formation of harmful free radicals through processes such as the Fenton reaction. These free radicals can damage cells, proteins, and DNA, leading to oxidative stress and various diseases.
Curcumin’s interaction with metal ions helps mitigate this problem. By binding to excess metal ions, curcumin reduces their availability to participate in harmful reactions. This metal-chelating property enhances its antioxidant activity, making it more effective in protecting cells from oxidative damage. As a result, curcumin is being studied for its potential role in preventing and managing conditions associated with oxidative stress, such as neurodegenerative diseases, cardiovascular disorders, and aging.
Another important aspect of curcumin-metal interactions is their influence on curcumin’s stability and solubility. When curcumin forms complexes with metal ions, its chemical properties can change. In some cases, these complexes exhibit improved stability and enhanced biological activity. For example, certain metal-curcumin complexes have shown greater resistance to degradation compared to free curcumin, making them more suitable for pharmaceutical applications.
Key features of curcumin’s interaction with metal ions include:
-
Chelation ability: Binds with metal ions through β-diketone and hydroxyl groups
-
Antioxidant enhancement: Reduces free radical formation by sequestering metals
-
Improved stability: Metal complexes may be more stable than free curcumin
-
Biological activity modulation: Alters therapeutic effects depending on the metal involved
-
Potential therapeutic use: Explored in treating diseases linked to metal imbalance
-
Environmental applications: Used in removing heavy metals from water
-
Structural flexibility: Allows formation of different types of metal complexes
The interaction between curcumin and metal ions is also being explored in the field of medicine, particularly in the treatment of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease. These conditions are often associated with abnormal metal accumulation in the brain. Curcumin’s ability to bind and regulate metal ions may help reduce toxicity and protect neural tissues. Additionally, some studies suggest that curcumin-metal complexes may have enhanced ability to cross biological barriers, further increasing their therapeutic potential.
In cancer research, curcumin-metal complexes are being investigated for their potential anti-cancer properties. Certain metal ions, when combined with curcumin, can form complexes that exhibit increased cytotoxicity toward cancer cells. These complexes may work by generating reactive oxygen species within cancer cells or by interfering with cellular processes essential for tumor growth. While this area of research is still developing, it highlights the versatility of curcumin as a platform for designing novel therapeutic agents.
Curcumin’s interaction with metal ions also has applications beyond medicine. In environmental science, it is being studied as a natural agent for removing heavy metals from contaminated water. The chelating ability of curcumin allows it to bind with toxic metals such as lead and cadmium, facilitating their removal. This makes it a promising candidate for eco-friendly water purification technologies.
Despite these advantages, there are challenges associated with curcumin-metal interactions. One concern is the variability in the stability and behavior of different metal-curcumin complexes. The nature of the metal ion, the pH of the environment, and other factors can influence the formation and properties of these complexes. This complexity requires careful study to ensure consistent and predictable outcomes, especially in pharmaceutical applications.
Another challenge is the limited bioavailability of curcumin itself. While forming complexes with metal ions may improve stability, it does not always guarantee better absorption in the body. Researchers are therefore exploring advanced delivery systems, such as nanoparticles and encapsulation techniques, to enhance the effectiveness of curcumin-metal complexes.
The study of curcumin’s interaction with metal ions also provides valuable insights into its broader chemical behavior. It demonstrates how natural compounds can engage in complex chemical processes that have significant biological and practical implications. This understanding not only enhances our knowledge of curcumin but also opens the door to new applications in science and technology.
In addition, the interaction of curcumin with essential metals such as zinc and magnesium may have beneficial effects on health. These metals play important roles in enzyme function and cellular processes, and their interaction with curcumin could influence how they are utilized in the body. However, more research is needed to fully understand these interactions and their implications.
In conclusion, the interaction of curcumin with metal ions is a fascinating and multifaceted area of study. Through its ability to chelate metals, curcumin enhances its antioxidant activity, improves stability, and opens up new possibilities for therapeutic and environmental applications. While challenges such as bioavailability and complex behavior remain, ongoing research continues to uncover the potential of curcumin-metal complexes. As our understanding deepens, this natural compound may play an increasingly important role in addressing some of the most pressing challenges in health, medicine, and environmental sustainability.