Molecular Structure and Functional Groups of Curcumin
Curcumin, the principal bioactive compound found in turmeric, has attracted significant attention in the fields of chemistry, pharmacology, and medicine. Responsible for the characteristic yellow color of turmeric, curcumin is more than just a pigment—it is a complex organic molecule with a unique structure that underpins its diverse biological activities. Understanding the molecular structure and functional groups of curcumin is essential for appreciating how it interacts with biological systems and why it holds such promise in therapeutic applications.
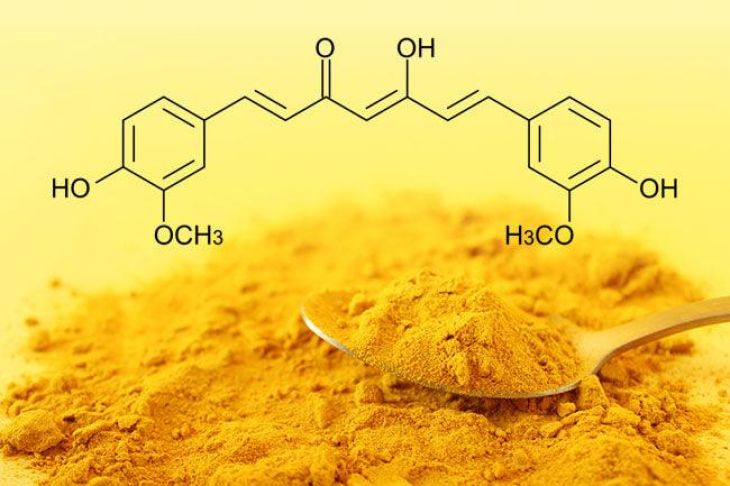
Chemically, curcumin belongs to a class of compounds known as polyphenols. Its systematic name is diferuloylmethane, reflecting its structure, which consists of two aromatic ring systems connected by a seven-carbon linker. This linker includes conjugated double bonds and a β-diketone moiety, both of which play critical roles in the compound’s chemical behavior and biological activity.
The molecular formula of curcumin is C₂₁H₂₀O₆, indicating that it is composed of carbon, hydrogen, and oxygen atoms arranged in a highly specific configuration. The structure is symmetrical, with two identical halves joined by a central chain. Each aromatic ring contains methoxy (-OCH₃) and hydroxyl (-OH) groups, which are key functional groups responsible for many of curcumin’s properties.
One of the most distinctive features of curcumin’s structure is its conjugated system of double bonds. This extended conjugation allows electrons to move freely across the molecule, contributing to its stability and its ability to absorb visible light. This is why curcumin appears bright yellow. More importantly, this conjugated system plays a crucial role in its antioxidant activity, enabling curcumin to neutralize free radicals by donating electrons.
The β-diketone group present in the central chain of curcumin is another important structural element. This group exists in equilibrium between two forms: the keto form and the enol form, a phenomenon known as tautomerism. The enol form is generally more stable in solution and contributes significantly to curcumin’s biological activity. This structural flexibility allows curcumin to interact with various molecular targets in the body.
Key functional groups present in curcumin include:
-
Phenolic hydroxyl (-OH) groups: Responsible for antioxidant activity and free radical scavenging
-
Methoxy (-OCH₃) groups: Contribute to chemical stability and influence solubility
-
β-diketone group: Plays a role in metal chelation and tautomerism
-
Conjugated double bonds: Provide structural stability and enable electron delocalization
-
Aromatic rings: Enhance interaction with biological molecules and membranes
The phenolic hydroxyl groups in curcumin are particularly important for its antioxidant properties. These groups can donate hydrogen atoms to neutralize reactive oxygen species, thereby protecting cells from oxidative damage. This mechanism is central to curcumin’s role in preventing chronic diseases and supporting overall health.
Methoxy groups, located on the aromatic rings, also play a significant role in curcumin’s chemical behavior. They influence the molecule’s polarity and solubility, which in turn affect its absorption and distribution in the body. These groups may also contribute to curcumin’s anti-inflammatory and antimicrobial activities by enhancing its interaction with specific enzymes and receptors.
The β-diketone moiety is known for its ability to bind metal ions, a property referred to as chelation. This is particularly relevant in biological systems, where metal ions such as iron and copper can catalyze the formation of harmful free radicals. By binding to these metals, curcumin can inhibit such reactions, further contributing to its antioxidant effects.
Another important aspect of curcumin’s structure is its lipophilicity, or its ability to dissolve in fats. This property allows curcumin to interact with cell membranes, which are primarily composed of lipids. As a result, curcumin can influence membrane structure and function, potentially affecting processes such as cell signaling and transport.
However, the same structural features that give curcumin its beneficial properties also contribute to its limitations. For instance, its poor water solubility and rapid metabolism in the body result in low bioavailability. This means that only a small fraction of consumed curcumin reaches the bloodstream in an active form. Researchers are actively working on modifying its structure or developing new formulations to overcome these challenges and enhance its therapeutic potential.
The study of curcumin’s molecular structure has also inspired the development of synthetic analogs. By altering specific functional groups or modifying the central linker, scientists aim to create compounds with improved stability, bioavailability, and biological activity. These analogs may offer new possibilities for drug development, particularly in the treatment of cancer, inflammation, and neurodegenerative diseases.
In addition to its biological applications, curcumin’s structure has made it a subject of interest in materials science. Its ability to absorb light and undergo chemical transformations has led to its use in sensors, dyes, and even experimental technologies. This highlights the versatility of curcumin as a molecule with both biological and industrial significance.
Understanding the relationship between structure and function is a fundamental concept in chemistry, and curcumin serves as an excellent example. Each functional group within the molecule contributes to its overall behavior, influencing how it interacts with other molecules and how it performs in biological systems. This interconnectedness underscores the importance of molecular design in both natural and synthetic compounds.
In conclusion, the molecular structure and functional groups of curcumin are central to its wide range of properties and applications. From its antioxidant and anti-inflammatory effects to its role in emerging scientific research, curcumin’s unique chemical architecture enables it to interact with biological systems in complex and beneficial ways. While challenges such as low bioavailability remain, ongoing research continues to explore innovative solutions. As our understanding of curcumin’s molecular structure deepens, it is likely to play an increasingly important role in science, medicine, and technology.