How Epigenetics Changes Gene Expression Without Altering DNA
The human body is made up of trillions of cells, and nearly every cell contains the same DNA sequence. This DNA acts as the biological blueprint for building and maintaining life. However, not all genes are active at the same time in every cell. For example, a liver cell behaves very differently from a brain cell, even though both contain identical genetic information. The reason behind this difference lies in the regulation of gene expression. One of the most fascinating mechanisms responsible for controlling how genes function is epigenetics.
Epigenetics refers to changes in gene activity that do not involve alterations to the underlying DNA sequence. Instead of changing the genetic code itself, epigenetic processes influence how genes are turned on or off. These modifications act like switches or dimmers that control the level of gene expression. Through epigenetics, cells can regulate which genes are active, how strongly they are expressed, and when they should be silent.
The term epigenetics literally means “above the genes.” It describes a layer of chemical modifications that sits on top of DNA and affects how genetic instructions are read by the cell. These changes can be influenced by various factors including age, environment, lifestyle, diet, and exposure to toxins. Because of this, epigenetics provides a powerful explanation for how environmental conditions can shape biological outcomes without modifying the DNA sequence itself.
One of the main mechanisms of epigenetic regulation involves chemical tags that attach to DNA or to proteins called histones, which help package DNA inside the nucleus. DNA in human cells is tightly wrapped around histone proteins to form a structure known as chromatin. When this chromatin structure is tightly packed, genes are generally inaccessible and remain inactive. When it is more loosely packed, genes become easier for the cell to read and can be actively expressed.
Among the different epigenetic processes, DNA methylation is one of the most widely studied. This process involves the addition of a small chemical group called a methyl group to specific regions of DNA. When methyl groups attach to gene promoter regions, they often suppress gene activity. In this way, DNA methylation acts like a molecular lock that prevents certain genes from being expressed.
Another important mechanism is histone modification. Histone proteins can undergo several chemical changes, including acetylation, methylation, phosphorylation, and ubiquitination. These modifications alter how tightly DNA is wrapped around histones. When histones are acetylated, the chromatin becomes more relaxed, allowing genes to be actively transcribed. In contrast, certain histone modifications can tighten DNA packing and reduce gene expression.
A third layer of epigenetic control involves non-coding RNA molecules. These RNA molecules do not produce proteins but instead regulate gene activity by interacting with DNA, messenger RNA, or chromatin-modifying enzymes. Through these interactions, non-coding RNAs can either enhance or suppress the expression of specific genes.
Several core mechanisms explain how epigenetic changes regulate gene expression:
- DNA methylation: Addition of methyl groups to DNA that typically suppresses gene activity.
- Histone modification: Chemical changes to histone proteins that alter how tightly DNA is packaged.
- Chromatin remodeling: Structural adjustments that make DNA more or less accessible for transcription.
- Non-coding RNA regulation: RNA molecules that influence gene expression without producing proteins.
- Environmental influence: Factors such as diet, stress, toxins, and lifestyle that can modify epigenetic patterns.
- Reversible gene control: Unlike genetic mutations, many epigenetic changes can be reversed.
One remarkable feature of epigenetic modifications is their ability to respond to environmental signals. For example, diet can influence epigenetic markers because certain nutrients provide chemical components needed for DNA methylation processes. Similarly, exposure to pollutants or chronic stress can alter epigenetic patterns and affect how genes behave. These changes may influence health outcomes, sometimes increasing the risk of diseases such as cancer, diabetes, and neurological disorders.
Epigenetics also plays a crucial role during development. When a fertilized egg begins dividing, all resulting cells initially have the potential to become any cell type in the body. As development progresses, epigenetic signals guide cells to specialize into different tissues such as muscle, nerve, or skin. By activating specific genes and silencing others, epigenetic mechanisms ensure that each cell develops its correct identity and function.
Another fascinating aspect of epigenetics is its potential role in inheritance. Some epigenetic changes can be passed from one generation to the next. Although most epigenetic marks are reset during reproduction, certain modifications may escape this reprogramming process. This means that environmental experiences, such as nutrition or stress, might influence the gene expression patterns of future generations. This concept is often referred to as transgenerational epigenetic inheritance.
Epigenetics has also become a major focus in medical research. Scientists have discovered that abnormal epigenetic changes can disrupt normal gene regulation and contribute to disease development. In cancer, for instance, tumor suppressor genes that normally prevent uncontrolled cell growth may become silenced through excessive DNA methylation. At the same time, genes that promote cell division may become abnormally activated. Understanding these epigenetic disruptions helps researchers identify new strategies for diagnosis and treatment.
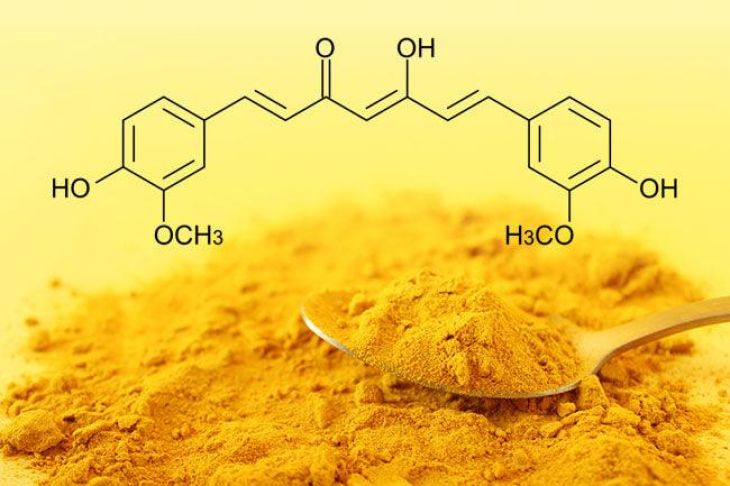
One promising area of research involves epigenetic therapy. Unlike traditional treatments that target DNA mutations directly, epigenetic therapies aim to reverse harmful epigenetic changes. Certain drugs can remove abnormal methyl groups or modify histone proteins, restoring normal gene expression patterns. Because epigenetic changes are often reversible, these therapies offer exciting possibilities for treating diseases that were previously difficult to manage.
In addition to disease treatment, epigenetic research is transforming our understanding of health and aging. As individuals grow older, their epigenetic patterns gradually change. Scientists have even developed “epigenetic clocks” that estimate biological age by analyzing specific DNA methylation markers. These discoveries may help researchers better understand aging processes and identify ways to promote healthier, longer lives.
In conclusion, epigenetics represents a powerful layer of biological regulation that determines how genes are expressed without altering the DNA sequence itself. Through mechanisms such as DNA methylation, histone modification, and non-coding RNA activity, cells can precisely control gene function in response to developmental signals and environmental influences. These epigenetic processes help explain how identical DNA can produce diverse cell types, how lifestyle factors affect health, and how certain diseases develop. As research in this field continues to grow, epigenetics is likely to play an increasingly important role in medicine, offering new insights into disease prevention, personalized treatment, and the complex relationship between genes and the environment.